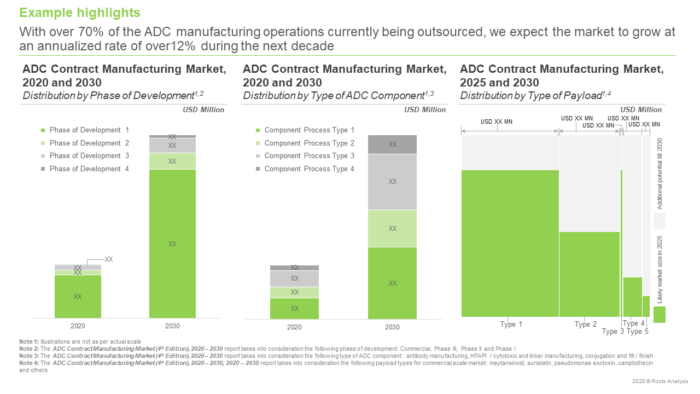
Since the success of ADCETRIS® (approved in 2011), antibody drug conjugates (ADCs) are now considered a versatile therapeutic tool and have been accepted into the contemporary portfolio of mainstream healthcare solutions. Over time, clinical researchers have been able to further their understanding of the intricacies of ADC design and have also improved the development process of these complex pharmacological interventions. Some of the recently approved ADC therapeutics include BLENREP® (2020), TRODELVYTM (2020), PadcevTM (2019) and POLIVY® (2019). In addition, there are close to 250 unique ADC product candidates under development. Several big pharma players, including AstraZeneca, GlaxoSmithKline, Pfizer, Roche and Takeda, have also acquired stake in this market. Moreover, the fact that companies involved in the development of ADCs, have received over USD 5 billion in capital investments (since 2011), attests to the therapeutic potential and growing popularity of this novel class of targeted therapeutics. However, the impending growth of the ADC therapeutics market highlights the rising importance of establishing advanced manufacturing capacities in order to meet the anticipated demand. Not all stakeholders in the industry possess end to end capabilities / infrastructure to support the design, development and manufacturing of these complex and highly potent pharmacological entities.